|
7/3/2023 0 Comments Keynote 590 Since the approval of trastuzumab in advanced metastatic GE cancer, the role of trastuzumab in resectable esophageal cancer has been debated in the absence of phase 3 randomized trial. Overexpression and/or amplification of HER2 occurs in 15% to 20% of GE cancers. This study showed a significant improvement in median disease-free survival (DFS) with the use of adjuvant nivolumab vs placebo (22.4 vs 11 months HR, 0.69 P <. The FDA recently approved the use of adjuvant nivolumab (Opdivo) for patients with stage II/III esophageal/GEJ cancer with residual pathologic disease after trimodality treatment regardless of PD-L1 status or histology, based on CheckMate 577 trial (NCT02743494). There is paucity of data regarding adjuvant treatment options in patients with residual disease post surgery. Even after trimodality therapy, there may be a high risk of recurrence for patients who do not achieve a pathologic complete response. Locally advanced esophageal and gastroesophageal junction (GEJ) cancers are aggressive tumor types that often require multiple approaches to address the disease, including neoadjuvant chemoradiotherapy and surgery. 2ĭevelopments for Resectable Esophageal Cancer Treatment The use of upfront comprehensive next-generation sequencing on tumor tissue is recommended it is not only associated with shorter time to test results, shorter time to treatment, but is also cost effective and eliminates the risk of missing rare but important oncogenes such as an NTRK fusion. Gastroesophageal (GE) cancers should be tested for HER2 amplification, microsatellite instability (MSI), PD-L1, tumor mutational burden (TMB), and NTRK fusion as these biomarkers can inform use of immune checkpoint inhibitor combinations, targeted agents as well as sequence of therapies. There have been significant advances in the treatment landscape for upper gastrointestinal cancers in the past year, with novel approaches available for patients with resectable disease and numerous targeted therapies now approved for advanced disease. 1Ī better understanding of the disease biology, identification of actionable genetic alterations and use of immunotherapy is heralding an era of precision therapy for treatment of esophageal cancers. 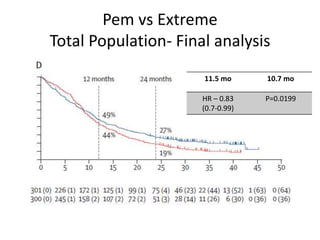
The histology can vary by region, for example high rates of esophageal adenocarcinoma are predominately seen in North America compared with squamous cell carcinoma, which is predominantly seen in Asia, Europe, and Africa. 
The 2 most common types of esophageal cancer types are squamous cell carcinoma and adenocarcinoma. This disease is the sixth most common cause of cancer-related deaths and the eighth most common cancer worldwide with a 5-year survival rate of less than 20%. 
The lifetime risk of esophageal cancer in the United States is about 1 in 125 in men and about 1 in 417 in women. In the United States, esophageal cancer makes up about 1% of all cancers and it is estimated that about 20,640 new people with esophageal cancer will be diagnosed in 2022. 
Nitika Sharma, MD, is a board-certified and fellowship-trained medical oncologist and hematologist at Cancer Treatment Centers of America, Atlanta.Īpril is Esophageal Cancer Awareness Month. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
